CharacteristicsPutty is a bone paste with at least 80% micronized heterologous bone (granulometry up to 300 µm) and collagen gel. It is made with an exclusive process that provides the product with exceptional malleability and plasticity, making it easy to apply into peri-implant defects with walls. Thanks to its collagen component, the product facilitates blood clotting and the subsequent invasion of repairing and regenerative cells, showing an osteoconductive behaviour(1).
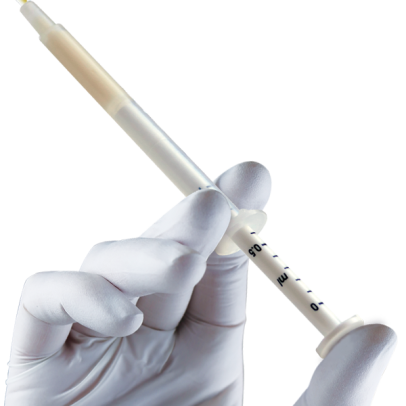
Successful grafting needs complete stability of the biomaterial: for this reason Putty must be used only in cavities able to firmly contain it. Therefore, Putty must not be grafted in two wall defects or in lateral access sinus lift procedures.HandlingInject the product and adapt it to defect morphology without compression; any non-stable residue must be removed before soft tissue suture.
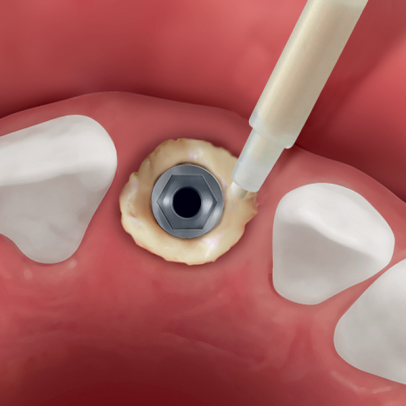
An Evolution membrane is recommended to protect Putty grafted in peri-implant defects.Clinical informationThe extraordinary handling properties of Putty syringe make this product the ideal choice for self-contained peri-implant defects(2) and all small defects that present a self-contained cavity. Furthermore, the Tecnoss® manufacturing process avoids granules ceramization, allowing a progressive resorption of the biomaterial and, at the same time, an adequate new-bone formation rate(3). Putty‘s “soft” consistency also guarantees an easy and healthy soft-tissues healing.
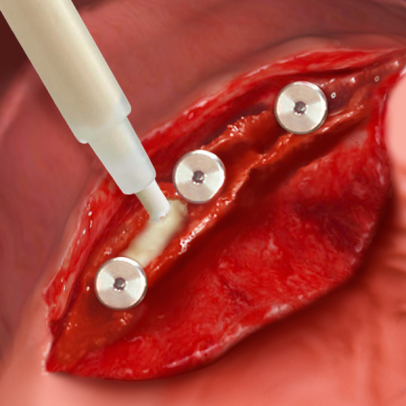
Thanks to these unique characteristics, Putty has been effectively used for peri-implant defects regeneration: following immediate post-extractive implants placement, Putty can be injected between the defect walls and the implant, guaranteeing a perfect filling of the entire defect volume(4). The product versatility also makes Putty the ideal solution when bone tissue has been lost due to peri-implantitis as long as the containing walls are present. In fact, the primary condition for gaining a successful regeneration is to achieve the biomaterial initial stability. Therefore, Putty must be used only in defects where the surrounding walls guarantee such condition: for example inside the bone crest when ridge-split technique is adopted(5), or with horizontally resorbed crests, in association with OsteoBiol® Lamina (Bone Layer technique)(6).
- Tissue origin :
Cortico-cancellous heterologous bone mix - Tissue collagen :
Preserved plus an additional 20% collagen gel - Physical form :
Plastic consistency composed of collagen gel loaded with 80% micronized bone mix - Composition :
80% granulated mix, 20% collagen gel - Granulometry :
Up to 300 µm - Re-entry time :
About 4 months - Packaging :
Syringe: 0.25 cc, 0.5 cc, 3×0.25 cc, 3×0.5 cc
Wide tip syringe: 1.0 cc - Product codes :
HPT52S | 1 Syringe | 0.25 cc | Porcine
HPT09S | 1 Syringe | 0.5 cc | Porcine
HPT09E | 1 Syringe | 0.5 cc | Equine
HPT32S | 3 Syringes | 3×0.25 cc | Porcine
HPT32E | 3 Syringes | 3×0.25 cc | Equine
HPT35S | 3 Syringes | 3×0.5 cc | Porcine
HPT35E | 3 Syringes | 3×0.5 cc | Equine
HPT61S | 1 Wide tip syringe | 1.0 cc | Porcine
HPT61E | 1 Wide tip syringes | 1.0 cc | Equine